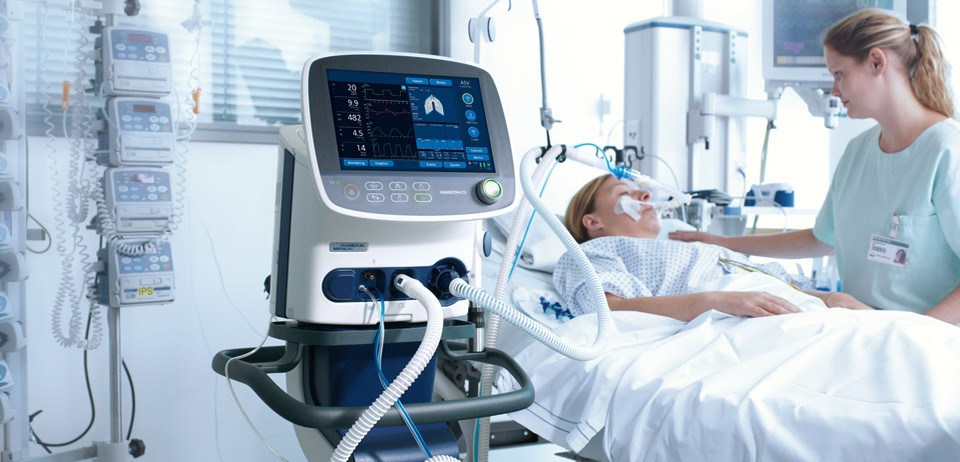
Technical inspection of the ventilator
This article explains why it is so important to perform technical inspections of ventilators and who can perform them. Medical devices, especially those that save lives and protect health, must be reliable – that is why the maintenance and servicing of medical devices is so important. A ventilator is a medical device that supports or replaces a patient's muscles in the breathing process. The device enables artificial, forced breathing, which is used in situations where breathing no longer functions (as a result of injury, illness or the use of drugs that block neuromuscular excitation) or to facilitate it (when the patient is breathing on their own but this function is difficult or does not meet the body's oxygen requirements due to the patient's condition).. Technical inspection of ventilators is carried out to ensure the continuity of operation of this device, which is extremely important for therapy. The inspection of the ventilator includes an electrical safety test, a functional test with measurements of functions and settings, maintenance of the device, and the possible replacement of operating parts and software updates. According to the Medical Devices Act of 20 May 2010, all medical devices, including ventilators, must undergo technical inspection. These can be carried out by ZTM INNOVATIONS or P&P CITO, among others.





 In 1995, the first diathermy model, KENTAMED 1, was launched and within a few years became the most popular coagulator in private clinics and practices in Bulgaria. In 2003 and 2010, KENTAMED achieved a breakthrough on the international markets with the second and third generations of its devices and is now represented in over 30 countries worldwide. At the beginning of 2010, the company launched the first model (KENTAMED RF-B) of the fourth generation. All products in this generation are based on Hartt solutions (Hardware Skorygowany Odpowiedź na Tissue Type). They are based on a series of precise load characteristics – separate for each model. In addition, these properties are confirmed by international certificates. Surgical diathermy devices are an important part of the equipment in most operating theatres and are now among the best-known and most frequently used devices by surgeons. This device is used in surgery for cutting with the aid of high-frequency electric current. Surgical diathermy devices differ from electrocautery devices in that both the cutting and coagulation effects can be achieved with a single device. This technique is used for:
In 1995, the first diathermy model, KENTAMED 1, was launched and within a few years became the most popular coagulator in private clinics and practices in Bulgaria. In 2003 and 2010, KENTAMED achieved a breakthrough on the international markets with the second and third generations of its devices and is now represented in over 30 countries worldwide. At the beginning of 2010, the company launched the first model (KENTAMED RF-B) of the fourth generation. All products in this generation are based on Hartt solutions (Hardware Skorygowany Odpowiedź na Tissue Type). They are based on a series of precise load characteristics – separate for each model. In addition, these properties are confirmed by international certificates. Surgical diathermy devices are an important part of the equipment in most operating theatres and are now among the best-known and most frequently used devices by surgeons. This device is used in surgery for cutting with the aid of high-frequency electric current. Surgical diathermy devices differ from electrocautery devices in that both the cutting and coagulation effects can be achieved with a single device. This technique is used for:  Surgical diathermy was first developed in 1926 by William Bovie and is based on the electrical generation of heat through the flow of high-frequency current through biological tissue. This technique allows the high-frequency current to cut or coagulate tissue, minimising blood loss and reducing operating time.
Surgical diathermy was first developed in 1926 by William Bovie and is based on the electrical generation of heat through the flow of high-frequency current through biological tissue. This technique allows the high-frequency current to cut or coagulate tissue, minimising blood loss and reducing operating time. 





